hey..
- Combustion of fuel is one of the major causes of air pollution in urban areas. Burning of fossil fuels such as natural gas and gasoline leads to the emission of carbon dioxide and other gases, which deteriorate the quality of air, making it polluted.
- Increased level of carbon dioxide in the atmosphere is one of the prime causes of air pollution. Power plants, exhaust fumes of automobiles, airplanes and other human activities involving the burning of gasoline and natural gas are related to the emission of this greenhouse gas.
- The chlorofluorocarbons (CFCs), a class of synthetic chemicals used in refrigerants and aerosol propellants, have caused hole in Earth's ozone layer. The use of this banned chemicals is related with the increasing levels of air pollution.
- Sulfur dioxide is one of the components of smog, which is related with contamination of the Earth's atmosphere. This synthetic chemical is the prime cause of acid rain.
- Air pollution is partially caused by the particulates formed by a variety of substances, such as dust, pollen and other organic materials.
- Increased road and air traffic is another reason related to the high level of air pollution.
- Asthma is a chronic disease common among children and adults. The shortness of breath, a prime symptom of this disease, can be triggered by pollutants present in the air, such as smog.
- Lung infections can occur when polluted air is inhaled. This can also lead to shortness of breath.
- Air pollution may influence the development of Chronic Obstructive Pulmonary Disease (COPD).
- Air pollution can also lead to lung cancer.
- Air pollution can result in blockage in the coronary artery or blood vessels that supply blood to the heart. This condition is diagnosed as Coronary Artery Disease.
- High level of pollution in the air can also lead to heart failure. Irregular or abnormal rhythms of heartbeat are often cited as health problems related to air pollution.
hope u got it..!! :)
- 8
AIR POLLUTION
Air Pollution, contamination of the atmosphere by gaseous, liquid, or solid wastes or by-products that can endanger human health and the health and welfare of plants and animals, or can attack materials, reduce visibility, or produce undesirable odours. Among air pollutants emitted by natural sources, only the radioactive gas radon is recognized as a widespread major health threat, although gases and particles from volcanic eruptions can cause serious more localized problems. A by-product of the radioactive decay of uranium minerals in certain kinds of rock, radon seeps into the basements of homes built on these rocks, posing a risk of lung cancer to residents.
Each year industrially developed countries generate billions of tons of pollutants. The most prevalent and widely dispersed air pollutants are described in the accompanying table. The level is usually given in terms of atmospheric concentrations (micrograms of pollutants per cubic metre of air) or, for gases, in terms of parts per million, that is, millilitres of gas per thousand litres of air. Many come from directly identifiable sources; sulphur dioxide, for example, comes from electric power plants burning coal or oil. Others are formed through the action of sunlight on previously emitted reactive materials (called precursors). For example, ozone, a dangerous pollutant in smog, is produced by the interaction of hydrocarbons and nitrogen oxides under the influence of sunlight. Ozone also causes serious crop damage. On the other hand, the discovery in the 1980s that air pollutants such as fluorocarbons are causing a loss of ozone from the Earth's protective ozone layer has caused the phasing out of these materials. A further category of air pollution is heavy metals, present as particulates and arising from many industrial processes.
| II | METEOROLOGY AND HEALTH EFFECTS |
Pollutant concentration is reduced by atmospheric mixing, which depends on such weather conditions as temperature, wind speed, and the movement of high and low pressure systems and their interaction with the local topography, for example, mountains and valleys. Normally, temperature decreases with altitude. But when a colder layer of air settles under a warm layer, producing a temperature or thermal inversion, atmospheric mixing is retarded and pollutants may accumulate near the ground. Inversions can become sustained under a stationary high-pressure system coupled with low wind speeds.
Periods of only three days of poor atmospheric mixing can lead to high concentrations of hazardous materials in high-pollution areas and, under severe conditions, can result in injury and even death. An inversion over Donora, Pennsylvania, in 1948 caused respiratory illness in over 6,000 people and led to the deaths of 20. Severe pollution in London took 3,500 to 4,000 lives in 1952 and another 700 in 1962. Release of methyl isocyanate into the air during a temperature inversion caused the disaster at Bhopal, India, in December 1984, with at least 3,300 deaths and more than 20,000 illnesses. The effects of long-term exposure to low concentrations are not well defined; however, those most at risk are the very young, the elderly, smokers, workers whose jobs expose them to toxic materials, and people with heart or lung disease. Other adverse effects of air pollution are injury to livestock and crops.
Often, the first noticeable effects of pollution are aesthetic and may not necessarily be dangerous. These include visibility reduction due to tiny particles suspended in air, or bad odours, such as the rotten egg smell produced by hydrogen sulphide emanating from pulp and paper mills.
| III | SOURCES AND CONTROL |
The combustion of coal, oil, and petrol accounts for much of the airborne pollutants. About 60 per cent of the sulphur dioxide and 20 per cent of the nitrogen oxides emitted into the atmosphere in the United Kingdom are produced by fossil-fuel-fired electric power plants. About 70 per cent of the carbon monoxide and 50 per cent of the nitrogen oxides come from burning petrol and diesel in cars and lorries. Other major pollution sources include iron and steel mills; smelters; municipal incinerators; oil refineries; cement plants; and nitric and sulphuric acid plants.
Potential pollutants may exist in the materials entering a chemical or combustion process (such as sulphur in coal), or they may be produced as a result of the process itself. Carbon monoxide, for example, is a typical product of internal-combustion engines. Methods for controlling air pollution include removing the hazardous material before it is used, removing the pollutant after it is formed, or altering the process so that the pollutant is not formed or occurs only at very low levels. Car exhaust pollutants can be controlled by burning the fuel as completely as possible, by recirculating fumes from fuel tank, carburettor, and crankcase, and by changing the engine exhaust to harmless substances in catalytic converters. Industrially emitted particulates may be trapped in cyclones, electrostatic precipitators, and filters. Pollutant gases can be collected in liquids or on solids, or incinerated into harmless substances.
| IV | LARGE-SCALE EFFECTS |
The tall smokestacks used by industries and utilities do not remove pollutants but simply boost them higher into the atmosphere, thereby reducing their concentration at the site. These pollutants may then be transported over large distances and produce adverse effects in areas far from the site of the original emission. Sulphur dioxide and nitrogen oxide emissions from Britain and other industrialized countries of Western and Central Europe are causing acid rain in Norway and Sweden. The pH level, or relative acidity, of many freshwater lakes has been altered so dramatically by acid rain that entire fish populations have been destroyed. Sulphur dioxide emissions and the subsequent formation of sulphuric acid can also be responsible for the attack on limestone and marble at large distances from the source. There are also claims that acid rain is causing widespread damage to forests in Europe, but the precise role is unclear and earlier predictions of large-scale forest death are unfounded.
The worldwide increase in the burning of coal and oil since the late 1940s has led to ever-increasing concentrations of carbon dioxide. The resulting “greenhouse effect”, which allows solar energy to enter the atmosphere but reduces the reemission of infrared radiation from the Earth, may well lead to a warming trend that might affect the global climate and lead to a partial melting of the polar ice-caps. Possibly an increase in cloud cover or absorption of excess carbon dioxide by the oceans (in the so-called carbon cycle) would check the greenhouse effect before it reached the stage of polar melting. Nevertheless, many research reports released during the 1990s have indicated that the greenhouse effect is definitely under way and that the nations of the world should be taking immediate steps to deal with it. In June 1999 a massive cloud of air pollution, roughly the size of the United States, was discovered 1 km to 3 km ( mi to 2 mi) above the Indian Ocean. The thick brown haze included soot, sulphates, nitrates, mineral dust, and significant amounts of gases such as carbon monoxide and sulphur dioxide. Scientists believe it was created by human activities, especially the burning of fossil fuels, and could have a significant impact on the regional and global climate, as well as plant and animal life.
| V | ACTION BY GOVERNMENTS |
Various countries have set standards in legislation in the form of concentration levels that are believed to be low enough to protect public health. Source emission standards are also specified to limit the discharge of pollutants into the air so that air-quality standards will be achieved. However, the nature of the problem requires the implementation of international environmental treaties, and to this end 49 countries agreed in March 1985 on a United Nations convention to protect the ozone layer. This “Montreal Protocol”, which was renegotiated in 1990 and 1992, called for the phaseout of certain chlorocarbons and fluorocarbons by the end of the century and provides aid to developing countries in making this transition. In addition, several international protocols have been aimed specifically at reducing the incidence of acid rain. In December 1999 the Montreal Protocol announced that almost all production and consumption of virgin ozone depleting substances had been phased out in the developed world. Similar control measures were introduced for developing countries in July 1999, and it was anticipated that all developing countries would be able to meet their freeze targets and subsequent obligations under the protocol.
Concern over trans-boundary air pollution, including acid rain, in Europe has led to the United Nations Economic Commission for Europe (UNECE) developing air quality guidelines, called Critical Loads and Levels, which represent thresholds below which it is believed that damage will not occur to different ecological systems. Critical Loads are based on the amount of acidity that an ecosystem can tolerate being deposited indefinitely. Critical Levels are concentrations of ozone, sulphur dioxide, and nitrogen oxides over different averaging times and applicable to different categories of vegetation. Massive cuts are being made in emissions in Europe in order to move towards these thresholds. In contrast, in many developing countries, pollutant concentrations are rising very rapidly due to increased industrialization and motor traffic; concern in such places is primarily with impacts on human health in cities. The World Health Organization has published air quality guidelines designed to protect health.
HOPE IT HELPS!
.
- 1
CAUSES:-
Air pollution is caused by a wide variety of things. The earth is great at cleaning the air on its own. However, air pollution has grown so much, the earth can no longer clean all of it. This is starting to have adverse effects on the environment such as causing acid rain, smog and a wide variety of health problems.
Combustion Engine Exhaust
Cars, trucks, jet airplanes and other combustion engine vehicles cause air pollution. The exhaust from these contains carbon monoxide, nitrous oxide and gaseous oxide. This type of air pollution creates smog (as seen in Los Angeles) which causes respiratory health problems and holes in the ozone layer, which increases the exposure to the sun's harmful rays.
Factories
Factories, office buildings, homes and power-generating stations burn fossil fuels, which cause air pollution. The burning of oil and coal (fossil fuels) also contributes to smog. This air pollution destroys plants, damages buildings and creates oxidation on iron.
Petroleum
Petroleum refineries release hydrocarbons and various particulates that pollute the air.
Power Lines
Some power lines are not insulated and are high voltage. This creates air pollution.
Pesticides
Pesticides used to kill indoor and outdoor pests, insecticides used to kill insects and herbicides use to kill weeds all cause air pollution.
Radioactive Fallout
Radioactive fallout causes air pollution from the nuclear energy dispersed, which is a dust.
Fertilizer Dust
Dust from fertilizers used to help plants grow causes air pollution.
Indoor Air Pollution
Sick building syndrome (SBS) is the term used when there is indoor air pollution. This happens when there is not enough ventilation to disburse the toxic fumes from new carpet, paint and/or cleaning chemicals that are used indoors. Mold can also cause SBS.
Mining Operations
Mining causes air pollution by releasing a variety of particles.
Mills and Plants
Mills and plants, include paper mills, chemical plants, iron mills, steel mills, cement plants and asphalt plants, release emissions into the air causing air pollution.
- 0
| ||||||||||||||||||||||||||||||||||||||||
- 0
The contribution of each source to local pollution will vary according to the type and number of local industrial processes, density and age of road transport and local weather conditions. Other pamphlets in this series will describe the problems in more detail and offer solutions for improvement. The figures used describing the respective production of pollutants by each sector are taken from "Air pollution in Europe" (EEA, 1997). Data relates to the year 1994 and to 28 countries in Europe(3). Siting fossil fuel power stations in mainly rural areas and distributing the pollution produced more evenly via tall chimneys has resulted in improved urban air quality, though they still remain a major source of pollution, mainly sulphur dioxide and nitrogen oxides. Better dispersion of pollutants emitted by tall chimneys leads to better dilution in the air and thus lower local concentrations of pollutants. This has however led to pollution being dispersed more widely and to transboundary air pollution. The countries of the European Union and those which are a party to the UNECE Convention on the Long Range Transport of Air Pollution, Second Sulphur Protocol, are committed to major reductions in sulphur dioxide emissions. Power generation is, however, likely to remain an important source of pollution for some time to come, particularly as some countries are reconsidering their programmes of nuclear power generation. Although fossil fuel power plants are the major source of industrial air pollution in many countries, all industry and many businesses, large and small, can be significant local sources of a wide range of air pollutants. The use of both regulatory and planning controls will help to minimise their effect on local air quality. All waste has the potential to affect the environment adversely by contaminating the air, soil or water. Poorly managed waste disposal sites (landfill or incineration) can also pose a danger to public health, through all these routes. Uncontrolled or poorly managed burning of waste (incineration) can result in the production of poisonous chemicals such as hydrochloric acid, dioxins, furans and heavy metals. Hydrochloric acid contributes locally to acid rain and is given off by the burning of plastics. If organic matter and plastics are burnt at low temperatures, dioxins and furans may be emitted. Modern, properly operated incinerators produce fumes which respect the strictest existing legislation. Air pollution from motor vehicles has, in many countries, replaced coal smoke as the major cause for concern; and the continuing growth in vehicle use means that efforts to reduce emissions from individual vehicles are in danger of being overtaken by increases in the volume of traffic. In much of eastern Europe the continued use of rather old cars, which are unable to meet modern pollution control requirements, means that efforts to control pollution from this source are going to be increasingly difficult. In those countries which have required the removal of lead from petrol, concentrations of lead in air from this source have been reduced to a level at which they are no longer a problem. Lead-free petrol has also made the use of "catalytic converters" possible. Catalysts substantially reduce emissions of hydrocarbons, NOx and carbon monoxide; they do however increase emissions of carbon dioxide, an important greenhouse gas, and have no effect on emissions of particles. Since 1993 all new petrol-engined cars in the European Union have to be fitted with catalytic converters. Prior to the introduction of cars fitted with catalytic converters, diesel-powered vehicles were considered "cleaner" than petrol-powered cars. EU legislation requires that they meet the same limits for hydrocarbons, NOx and CO as petrol-driven cars. Diesel fuel contains no lead but is a considerable source of particulate matter, PAHs and SO2. The introduction of lower sulphur diesel fuels throughout the EU will reduce emissions from this source (see table). In many countries, there has been a policy of progressively tightening emission standards for cars and lorries in line with EU directives and UNECE standards. However, much more will need to be done to ensure that reductions in vehicle emissions are not offset by the rapid increase in vehicle ownership and use. This is an area in which action by local authorities can make a significant impact on local air quality and indeed benefit the local community in terms both of their health and of local amenities. Each local authority will need to consider how it can best tackle the problem, bearing in mind the resources available and other priorities for cutting pollution. Improved public transport, "park and ride" schemes, traffic restrictions, planning guidelines and encouragement to cycle and walk are some of the measures that local authorities can take. Requiring vehicle owners (and businesses) to maintain their vehicles regularly will ensure that fuel is burnt efficiently and economically, and will therefore be less polluting; and fuel consumption is more efficient at lower speeds (60 - 90 km/h). Before about 1960, the domestic use of coal was the major source of particles. Concentrations of airborne particles in many European cities frequently exceeded 1000 µg/m3 and annual average concentrations of several hundred µg/m3 were commonplace. Today, annual average concentrations in most European cities have fallen to less than 30 µg/m3. In eastern Europe much higher concentrations still occur as, to a lesser extent, they do in southern European cities such as Athens. Brown coal (lignite) is a key source of particles in many parts of eastern Europe. Coal varies in composition and calorific value from mine to mine. Lignite is probably the poorest quality in terms of calorific value and generates most pollutants when used for domestic heating. Lignite contains 67% carbon (compared with the 95% in anthracite) and burns easily, though inefficiently, on an open fire. Special devices with carefully controlled air supplies are needed to burn anthracite but combustion is efficient and far less black smoke is produced. Conversion of open fires to stoves suitable for burning anthracite (or other smokeless fuel) should be considered by any local authority where coal smoke is a problem. The greater efficiency of controlled anthracite burning leads to a saving in overall fuel costs, though an initial investment in the necessary equipment must be made. Conversion to stoves which ensure complete combustion are also a possibility as are district heating schemes, using combined heat and power plants. The greater use of renewable energy (wind, solar, tidal, wave, etc.) and enhanced energy efficiency measures in homes and offices also helps improve air quality. Other important domestic sources of air pollution are:![]() Industrial Sources
Industrial Sources![]() Power Generation
Power Generation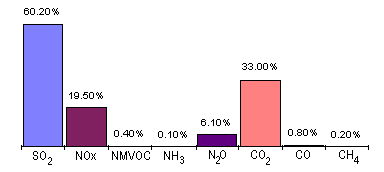
 Stricter operating practices and the use of modern abatement techniques have resulted in a considerable reduction in the amount of pollutants emitted from power stations; high concentrations do however occur in many eastern European countries, particularly from older power stations and from the use of high sulphur lignite or brown coal. Exceptional concentrations may also occur on a very local basis if a plume of smoke from, for example, an industrial chimney falls to the ground due to local atmospheric conditions.
Stricter operating practices and the use of modern abatement techniques have resulted in a considerable reduction in the amount of pollutants emitted from power stations; high concentrations do however occur in many eastern European countries, particularly from older power stations and from the use of high sulphur lignite or brown coal. Exceptional concentrations may also occur on a very local basis if a plume of smoke from, for example, an industrial chimney falls to the ground due to local atmospheric conditions.![]() Other Industry and waste disposal
Other Industry and waste disposal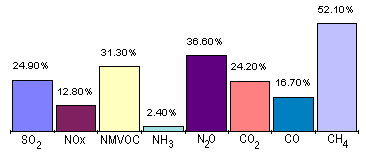
 Landfill and incineration are the two most common methods of waste disposal. If not properly managed landfill sites can cause a number of problems; these include the production of potentially explosive levels of methane gas (65%), dangerous levels of carbon dioxide (35%), plus trace concentrations of a range of organic gases and vapours. Landfill sites also have the potential to cause major odour when badly managed.
Landfill and incineration are the two most common methods of waste disposal. If not properly managed landfill sites can cause a number of problems; these include the production of potentially explosive levels of methane gas (65%), dangerous levels of carbon dioxide (35%), plus trace concentrations of a range of organic gases and vapours. Landfill sites also have the potential to cause major odour when badly managed.![]() Road Transport
Road Transport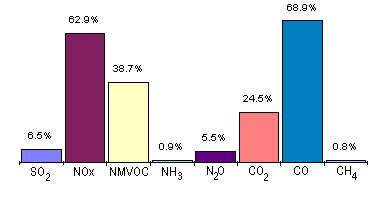
 The air pollutants produced as a result of the use of motor vehicles present a two-stage problem: primary and secondary pollutants. Primary pollutants produced by petrol-powered vehicles include carbon monoxide, nitric oxide, benzene, particulate matter and lead. Much of the lead emitted by vehicles burning leaded petrol emerges as particles. Diesel engines burn fuel in excess of air and so produce little carbon monoxide but, instead large quantities of carbon dioxide, (see table). Secondary pollutants produced as a result of the use of petrol-engined vehicles include nitrogen dioxide and ozone.
The air pollutants produced as a result of the use of motor vehicles present a two-stage problem: primary and secondary pollutants. Primary pollutants produced by petrol-powered vehicles include carbon monoxide, nitric oxide, benzene, particulate matter and lead. Much of the lead emitted by vehicles burning leaded petrol emerges as particles. Diesel engines burn fuel in excess of air and so produce little carbon monoxide but, instead large quantities of carbon dioxide, (see table). Secondary pollutants produced as a result of the use of petrol-engined vehicles include nitrogen dioxide and ozone.Measures for inclusion in a Local Transport Strategy Emission factor Environmental third class diesel Environmental first class diesel Ethanol Regulated pollutants: Carbon monoxide, g/km 3.3 1.9 0.25 Hydrocarbons, g/km 1.3 0.72 0.13 NOX, g/km 13.2 9.7 9.5 Particulates, mg/km 510 200 53 Unregulated pollutants: Carbon dioxide, kg/km 1.2 1.1 1.2 Formaldehyde, mg/km 70 29 18 Acetaldehyde, mg/km 19 20 72 Sum, particle-bound PAH (> 3 aromatic rings) mg/km 220 39 6.0 All emission factors from emission tests performed using the Braunschweig transient bus cycle. (From: Westerholm, R. and Wijk, A., "Vehicle exhausts", Swedish Environmental Protection Agency, Report 4528, Stockholm, 1996). ![]() Domestic Sources
Domestic Sources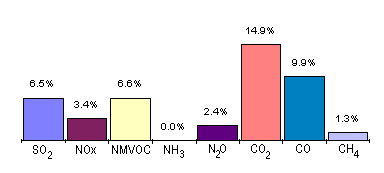
![]() Agriculture
Agriculture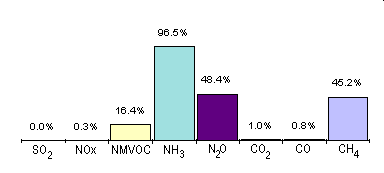
- 0

